Hydroxychloroquine retinal toxicity
A 22-year‐old Asian female was referred by her optometrist to the Centre for Eye Health, UNSW for a macular examination. She has been taking 100-200mg/day of hydroxychloroquine (Plaquenil) for systemic lupus erythematosus for nine years. She had no previous history of visual difficulty and denied any symptoms of blurred vision or any other visual disturbance at the time of consultation.
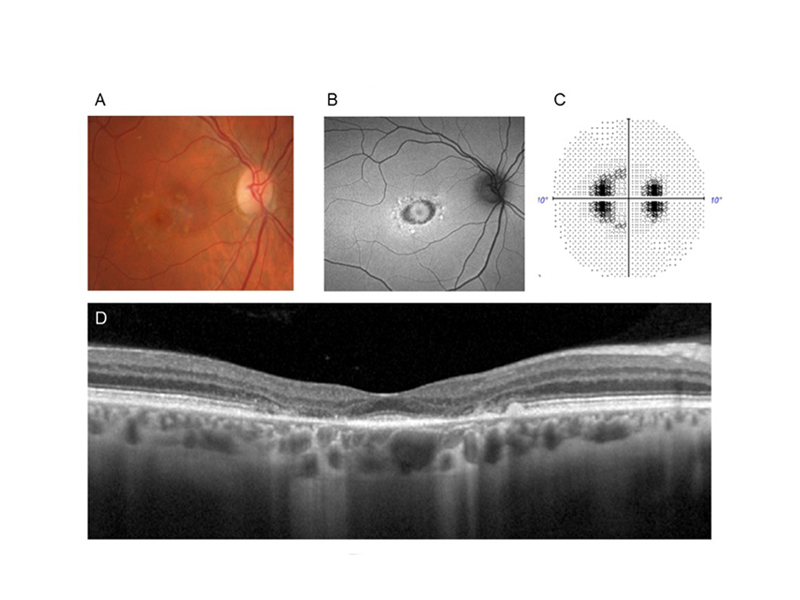
On examination, best‐corrected visual acuities were 6/6 in each eye. Contrast sensitivity measured 1.60 log units OD and 1.68 log units OS using the Mars test (normal range 1.72 to 1.92 log units). Anterior segment examination was unremarkable. Applanation intraocular pressures were 15mmHg OD and 18mmHg OS. Case images and results from the right eye are presented in Figure 1. Fundus examination (Figure 1A) and fundus autofluorescence (Figure 1B) were normal. Humphrey 30-2 SITA standard perimetry showed clustered depressions in the temporal side, mostly outside of the central ten degrees (Figure 1C). Spectral domain optical coherence tomography (SD-OCT) showed appreciable thinning of the outer nuclear layer and loss of the ellipsoid zone (EZ) in the temporal parafovea (Figure 1D) and inferonasal arcades (Figure 1E). The results from the left eye were similar and are omitted for brevity.
Which of the following tests is NOT recommended for screening hydroxychloroquine retinal toxicity?
- a. Colour fundus photography
- b. Spectral domain optical coherence tomography
- c. Fundus autofluorescence
- d. Automated visual fields
Diagnosis
Early hydroxychloroquine retinopathy
Answer
a. colour fundus photography is NOT recommended as a screening test for hydroxychloroquine retinopathy.
Hydroxychloroquine is a widely used drug for the treatment of autoimmune and inflammatory diseases. It is known to cause retinal damage and can potentially lead to vision loss in patients on long-term therapy.1 The classic presentation of hydroxychloroquine toxicity displays parafoveal loss of photoreceptors and RPE in a bull’s eye pattern, known as Bull’s eye maculopathy (Figure 2).2 However, this overt presentation implies late-stage toxicity by which vision loss has already occurred and the changes are not only irreversible but progressive despite drug discontinuation.3-5
Figure 1. Case images and field result from the patient’s right eye. A) Colour fundus photograph showing normal findings. B) Fundus autofluorescence similarly not showing any marked RPE abnormalities. C) 30-2 visual field result showing clustered depressions predominantly outside the central ten degrees. D) Spectral-domain optical coherence tomography (SD-OCT) showing temporal parafoveal thinning and disruption of inner segment ellipsoid zone. E) A second SD-OCT B-scan taken through the inferior posterior pole showing ellipsoid zone loss at the inferonasal vascular arcades.

Figure 2. Images from another case seen at the Centre with severe hydroxychloroquine toxicity (bull’s eye maculopathy). A) Bull’s eye paracentral pattern of pigmentary abnormalities in the colour fundus image. B) Autofluorescence shows RPE abnormality at the central macula. C) 10-2 visual field greyscale shows a partial ring scotoma. D) Spectral domain-OCT shows marked loss of parafoveal outer retina and RPE with homogenous signal hyper-transmission into the underlying choroid. Other images and the visual field results of this patient have been reported elsewhere.6
Screening for hydroxychloroquine retinal toxicity aims to detect signs at an early stage — before RPE loss— to prevent central vision loss. Frequently, the early toxicity exhibits no visible fundus lesions, no or minimal autofluorescence abnormality (due to the sparing of the RPE) and isolated and subtle signs on SD-OCT, which renders detection challenging. To further complicate matters, not all patients with hydroxychloroquine retinopathy display the classic parafoveal pattern; those with Asian ancestry typically display early pericentral damage (retinal changes 8 degrees from the fovea) near the vascular arcade (Figure 1).7
To enable the accurate detection of the early changes, clinicians should look for loss of the ellipsoid zone, parafoveal thinning of the outer nuclear layer on SD-OCT and corresponding functional changes on visual fields using a widefield structural and functional approach in such patients1 and be cognisant that these changes may lie in the extramacular location. SD-OCT and automated visual field tests are recommended as primary tests for screening toxicity, with FAF as a useful adjunctive test.8 FAF is particularly useful for displaying RPE changes, and may show increased autofluorescence in the early stage which becomes mottled as the damage to the RPE become more established.9 Amsler grid, and colour vision tests are not recommended for screening as they lack the reliability, sensitivity and specificity to detect early changes.8 Similarly, colour fundus photography is not recommended due to the low sensitivity to early damage.8
Patient outcome
The patient was referred to a retinal specialist and a copy of the report was forwarded to her general practitioner and rheumatologist.
What is known on this topic
- Hydroxychloroquine retinopathy is uncommon at recommended doses; however, the risk of toxicity rises significantly over time, affecting 20% after 20 years of use.8
- Risks for development of hydroxychloroquine retinopathy includes high dose (>5.0 mg/kg real weight), duration of use (>5 years), renal disease, Tamoxifen use and presence of macular disease.8
- Baseline evaluation should be performed within one year of commencing hydroxychloroquine therapy and annually after five years in patients without significant risk factors.8
- Visual acuity is a poor indicator of the severity of retinal toxicity. SD-OCT and visual fields are recommended to stage severity.1
- The pattern of toxicity can differ by race with pericentral damage more common in Asian patients.7
- Early detection of toxicity and discontinuation of therapy is crucial (involving care co-ordination among multiple health care practitioners) in limiting progression before vision is significantly affected.
What this case study illuminates
- Screening tests for hydroxychloroquine retinopathy should be adjusted based on race.8, 10 In Asian patients, 24-2 or 30-2 visual fields should be adopted instead of 10-2 and wide-field OCT and fundus autofluorescence should be acquired to facilitate the recognition of pericentral abnormalities.
What evidence gaps remain?
- The pathogenesis of hydroxychloroquine retinal toxicity is unclear and the reason for progressive retinopathy despite drug discontinuation remains elusive. 10
- The search for more reliable, objective, and sensitive screening tests is ongoing. Microperimetry has shown comparable sensitivity and specificity as multifocal electroretinogram – the gold standard for diagnosing retinal toxicity, although is yet to be widely adopted in a clinical setting.11
Author
Pauline Xu, BOptom (Hons), MOptom, GradCertOcTher
Lead clinical (Retina), Centre for Eye Health
Pauline received her undergraduate and Masters degree from UNSW. Before joining the Centre for Eye Health (CFEH), she practised full-scope optometry in both rural and urban practices followed by five years working with the Brien Holden Vision Institute as a research optometrist during which time she was involved in the production of several papers published in leading journals. Pauline is involved in the clinical supervision, lecturing of undergraduate and postgraduate optometry students through the School of Optometry and Vision Science at UNSW, has presented both webinars and workshops for CFEH and has also been invited to speak at national conferences. Through her work at CFEH she has developed a strong interest in inherited retinal and macular dystrophies.
Acknowledgements
The author thanks Angelica Ly for reviewing the manuscript.
Disclosures
The Centre for Eye Health receives primary funding from Guide Dogs NSW/ACT.
References
- 1. Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol 2014;132:1453-1460.
- 2. Geamănu Pancă A, Popa-Cherecheanu A, Marinescu B, Geamănu CD, Voinea LM. Retinal toxicity associated with chronic exposure to hydroxychloroquine and its ocular screening. Review. Journal of medicine and life 2014;7:322-326.
- 3. Marmor MF, Hu J. Effect of disease stage on progression of hydroxychloroquine retinopathy. JAMA Ophthalmol 2014;132:1105-1112.
- 4. Lally DR, Heier JS, Baumal C, et al. Expanded spectral domain-OCT findings in the early detection of hydroxychloroquine retinopathy and changes following drug cessation. Int J Retina Vitreous 2016;2:18.
- 5. Mititelu M, Wong BJ, Brenner M, Bryar PJ, Jampol LM, Fawzi AA. Progression of Hydroxychloroquine Toxic Effects After Drug Therapy Cessation: New Evidence From Multimodal Imaging. JAMA Ophthalmology 2013;131:1187-1197.
- 6. Xu P. Demystifying Bull’s Eye Maculopathy. Pharma 2020.
- 7. Melles RB, Marmor MF. Pericentral retinopathy and racial differences in hydroxychloroquine toxicity. Ophthalmology 2015;122:110-116.
- 8. Marmor MF, Kellner U, Lai TY, Melles RB, Mieler WF, American Academy of O. Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy (2016 Revision). Ophthalmology 2016;123:1386-1394.
- 9. Kellner U, Renner AB, Tillack H. Fundus autofluorescence and mfERG for early detection of retinal alterations in patients using chloroquine/hydroxychloroquine. Invest Ophthalmol Vis Sci 2006;47:3531-3538.
- 10. Yusuf IH, Sharma S, Luqmani R, Downes SM. Hydroxychloroquine retinopathy. Eye (Lond) 2017;31:828-845.
- 11. Iftikhar M, Kaur R, Nefalar A, et al. Microperimetry as a screening test for hydroxychloroquine retinopathy: The Hard-Risk-1 Study. Retina 2019;39:485-491.















