Case
A 14-year-old male was referred to the Centre for Eye Health, University of New South Wales for a macular assessment. The referring practitioner reported a white-yellow macular lesion in the left eye with suspicions of an inherited condition, given the patient’s father had a long history of vision loss. There was no history of eye injuries or other ocular conditions and he was in good general health.
On examination, best corrected visual acuities were 6/3.8 R and 6/15+1 L with no improvement with pinhole L. Amsler grid was described as normal in the right eye and generally blurry in the left eye. Contrast sensitivity was reduced, particularly in the left eye, at 1.56 log units R and 1.28 log units L with Mars testing (normal range 1.72 to 1.92 log units). Anterior eye examination was unremarkable. Dilated fundus examination revealed a healthy appearing right macula and a heterogeneous, white-yellow fibrotic nodule at the left macula with a possible overlying thin hemorrhage (Figure 1A). Fundus autofluorescence imaging revealed corresponding patchy autofluorescence (Figure 1B).1 Optical coherence tomography (OCT) through the left lesion showed a hyper-reflective, subretinal pigment epithelium (sub-RPE) fibrotic nodule, with associated elevation of the neurosensory retina and surrounding subretinal fluid (Figure 1C). OCT angiography of the avascular slab revealed abnormal vasculature suspicious of choroidal neovascularisation (Figure 1D).
Pictured is a colour fundus photograph showing a white-yellow lesion with superimposed OCT angiography findings. What do the flow signals on OCT angiography represent?
- a. Intraretinal hemorrhage
- b. Choroidal neovascularisation
- c. Chorioretinal atrophy
- d. Projection artefact
Diagnosis
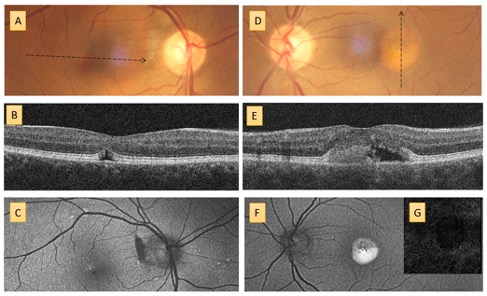
The presentation is consistent with Best’s Vitelliform Macula Dystrophy (BVMD). In light of his son’s clinical findings, the patient’s father pursued further assessment at the Centre for Eye Health. His habitual visual acuities were 6/12 R and 6/15+1 L with pinhole improvement of 6/9+2 R and 6/12+1 L. The imaging results are displayed below (Figure 2) and display classic changes consistent with BVMD.
Answer
b. Choroidal neovascuarisation

Best’s Vitelliform Macula Dystrophy (BVMD) is a progressive inherited macular dystrophy classically characterised by variable, bilateral deposition of yellowish, vitelliform material often attributed to lipofuscin in the RPE, a byproduct of photoreceptor outer-segment metabolism.1-3 In its classical manifestation, it involves a well circumscribed, dome-shaped, yellow macular lesion – commonly described as egg-yolk in appearance. The condition was first described by a German ophthalmologist, Frederich Best, in 1905 who reported the pedigree of an affected family. However, since then, a spectrum of other retinal degenerations caused by mutations in the BEST1 gene have been observed, known collectively as the bestrophinopathies.4 Best vitelliform macular dystrophy and adult-onset vitelliform macular dystrophy (AOVMD) are the most common phenotypes; are typically confined to the macular region and share an autosomal dominant inheritance pattern. The age of onset for BVMD is typically within the first two decades of life, which contrasts with AOVMD, as suggested by the name, with typical age of onset after fourth decade.5
Demonstrated herein, the phenotypic appearance of Best disease varies by disease stage, expressivity and penetrance.3 Five stages have been identified:2
Subclinical (stage 1) – evident in patients who carry the gene mutation but show no biomicroscopically detectable macular abnormalities. Such cases may demonstrate early electro-oculogram, outer retinal and choriocapillaris abnormalities.6
The vitelliform (stage 2) – with accumulation of lipofuscin and vitelliform material which is associated with relatively good preservation of visual acuity.7
This is followed by progressive degradation of vitelliform material and accumulation of subretinal fluid (commonly associated decrease in vision) and inferior accumulation of the vitelliform lesion, courtesy of gravity. This stage is often described as pseudohypopyon (stage 3) (Fig 2 D).7, 8
With the natural history of the disease, subretinal accumulations begin to decrease progressively associated with decreasing lesion height and further reduction in vision commonly known as vitelliruptive (stage 4).8
The end, atrophic/cicatricial stage (stage 5) of the disease involves atrophy of underlying retina with associated significant vision loss. Such as in the case described, a subset of patients develop subretinal fibrous deposition, and haemorrhagic detachment of the macula.
A more recent classification based on OCT findings has been proposed simplifying the stages to subclinical, vitelliform and atrophic.7 In this proposed classification, subclinical stage corresponds to limited outer retinal disruptions on OCT with no evidence of vitelliform material or subretinal fluid. Stages 2, 3 and 4 of the traditional classification are collapsed together into a collective vitelliform stage, which indicates outer retinal disruptions, vitelliform material and evidence of subretinal fluid. The atrophic stage is characterised by deeper alterations at the outer retinal level with limited vitelliform material and subretinal fluid. With increasing accessibility to multimodal imaging, this classification may be more widely adopted in the future.7
Patient outcome
The patient was referred to an ophthalmologist within one week. Fluorescein angiography was performed showing leakage at the left macula. He was diagnosed with left Best’s vitelliform macular disease complicated by a choroidal neovascular membrane and recommended left intravitreal anti-VEGF injection of Lucentis (ranibizumab) with the aim to cease macular exudation and preserve vision.
What is known on this topic
- While the nature of vision loss in BVMD is progressive and slow, cases with sudden decrease in vision associated with choroidal neovascularisation have been reported in the literature.9, 10
- Although traditionally considered rare,11 with multimodal imaging, particularly OCT angiography, choroidal neovascularisation is now considered to be an under-detected entity with up to 65 per cent of eyes in various stage of BVMD showing signs of choroidal neovascularisation (either exudative or non-exudative).12 Interestingly, natural history and development of these membranes follow a more benign course compared to age related macular degeneration.13
- Although, there is no consensus as to the treatment of choroidal neovascularisation in Best’s Vitelliform Macula Dystrophy, peer-reviewed case reports show CNV regression, reduction of subretinal fluid and improvement in visual acuity14, 15 with ranibizumab injections and no signs of new complications during the three-year follow-up. Similar successful case reports have been shown with other anti-VEGF injections.16
What this case illuminates
- Unilateral presentation of Best’s Vitelliform disease with secondary complication of choroidal neovascularisation in a young patient.
- Confirmation of the inheritance with the father displaying classic bilateral BVMD without the CNV complication his son displayed.
- Emerging technologies, in particular OCT angiography, and multimodal imaging have significant importance in detecting choroidal neovascularisation early without invasive techniques such as fluorescein angiography (particularly in a paediatric population). In some cases, it has proven to be superior17 and less challenging than conventional angiography particularly given presence of pre-existing subretinal deposits which stain on fluorescein angiography and can mislead the diagnosis.13
What evidence gaps remain?
- Although case reports showing the successful treatment of Best vitelliform macular dystrophy associated choroidal neovascularisation with anti-VEGF injections exist, there have been no large-scale randomised controlled trials which compare the outcomes to observation alone.13
- Further genetic studies to identify those at risk of complications is necessary.
Author
Meri Galoyan BOptom (Hons.), BSc
Staff Optometrist, Centre for Eye Health
Meri completed her BOptom / BSc (Hons) at University of New South Wales. She has found her passion for ocular pathology through her work at the Centre for Eye Health. She is actively involved in clinical services, student supervision and teaching. She has been involved in numerous webinars both within the Centre and nationally.
Acknowledgements
The author thanks Dr Angelica Ly, Pauline Xu, and Professor Michael Kalloniatis for reviewing the manuscript.
Disclosures
The Centre for Eye Health receives primary funding from Guide Dogs NSW/ACT.
References
1. Parodi MB, Iacono P, Campa C, Del Turco C, Bandello F. Fundus Autofluorescence Patterns in Best Vitelliform Macular Dystrophy. American Journal of Ophthalmology. 2014;158(5):1086-92.e2.
2. Gass JDM. Stereoscopic atlas of macular diseases. 4 ed. St. Louis: Mosby; 1997.
3. Spaide RF, Noble K, Morgan A, Freund KB. Vitelliform macular dystrophy. Ophthalmology. 2006;113(8):1392-400.
4. Toto L, Boon CJ, Di Antonio L, Battaglia Parodi M, Mastropasqua R, Antonucci I, et al. BESTROPHINOPATHY: A Spectrum of Ocular Abnormalities Caused by the c.614T>C Mutation in the BEST1 Gene. Retina. 2016;36(8):1586-95.
5. Chowers I, Tiosano L, Audo I, Grunin M, Boon CJ. Adult-onset foveomacular vitelliform dystrophy: A fresh perspective. Progress in retinal and eye research. 2015;47:64-85.
6. Parodi MB, Arrigo A, Calamuneri A, Aragona E, Bandello F. Multimodal imaging in subclinical best vitelliform macular dystrophy. The British journal of ophthalmology. 2020.
7. Battaglia Parodi M, Iacono P, Romano F, Bandello F. SPECTRAL DOMAIN OPTICAL COHERENCE TOMOGRAPHY FEATURES IN DIFFERENT STAGES OF BEST VITELLIFORM MACULAR DYSTROPHY. Retina. 2018;38(5):1041-6.
8. Battaglia Parodi M, Romano F, Arrigo A, Di Nunzio C, Buzzotta A, Alto G, et al. Natural course of the vitelliform stage in best vitelliform macular dystrophy: a five-year follow-up study. Graefe’s archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2020;258(2):297-301.
9. Alisa-Victoria K, Jin-Poi T, Shatriah I, Zunaina E, Ngah NF. Choroidal neovascularization secondary to Best’s vitelliform macular dystrophy in two siblings of a Malay family. Clin Ophthalmol. 2014;8:537-42.
10. Frennesson CI, Wadelius C, Nilsson SE. Best vitelliform macular dystrophy in a Swedish family: genetic analysis and a seven-year follow-up of photodynamic treatment of a young boy with choroidal neovascularization. Acta ophthalmologica. 2014;92(3):238-42.
11. Miller SA, Bresnick GH, Chandra SR. Choroidal neovascular membrane in Best’s vitelliform macular dystrophy. Am J Ophthalmol. 1976;82(2):252-5.
12. Parodi MB, Arrigo A, Bandello F. Optical Coherence Tomography Angiography Quantitative Assessment of Macular Neovascularization in Best Vitelliform Macular Dystrophy. Investigative ophthalmology & visual science. 2020;61(6):61.
13. Khan KN, Mahroo OA, Islam F, Webster AR, Moore AT, Michaelides M. FUNCTIONAL AND ANATOMICAL OUTCOMES OF CHOROIDAL NEOVASCULARIZATION COMPLICATING BEST1-RELATED RETINOPATHY. RETINA. 2017;37(7).
14. Batioglu F, Ozmert E, Suren E, Demirel S. Intravitreal Ranibizumab for the Treatment of Choroidal Neovascularization in Best’s Vitelliform Macular Dystrophy2012.
15. Ruiz-Moreno O, Calvo P, Ferrández B, Torrón C. Long-term outcomes of intravitreal ranibizumab for choroidal neovascularization secondary to Best’s disease: 3-year follow-up. Acta ophthalmologica. 2012;90(7):e574-5.
16. Celea C, Pop M, Avidis-Zamfiroiu N, Celea C. Evolution of Choroidal Neovascular Membrane in Best Disease after Single Intravitreal Bevacizumab. Case Report. Maedica. 2015;10(1):61-4.
17. Stattin M, Ahmed D, Glittenberg C, Krebs I, Ansari-Shahrezaei S. OPTICAL COHERENCE TOMOGRAPHY ANGIOGRAPHY FOR THE DETECTION OF SECONDARY CHOROIDAL NEOVASCULARIZATION IN VITELLIFORM MACULAR DYSTROPHY. Retinal cases & brief reports. 2020;14(1):49-52.
You might also like…
Hidden in plain sight
A case study on hydroxychloroquine retinal toxicity from the Centre for Eye Health.















